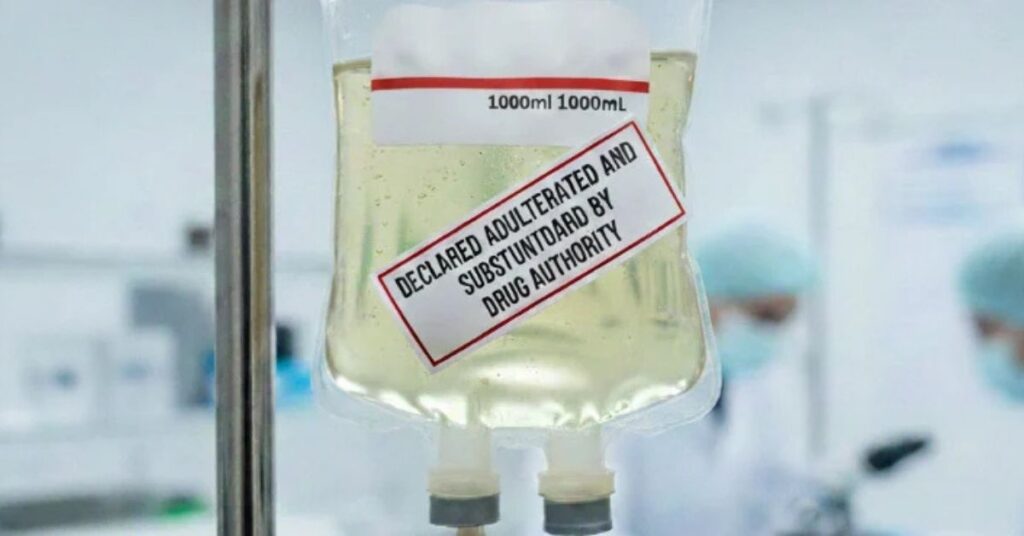
LAHORE (Kashmir English): The Directorate of Drugs Control, Punjab, has ordered the manufacturers to recall the declared substandard and adulterated batches of medicines from the market.
According to a public notice, the Drug Testing Laboratories Punjab declared the listed medicines as substandard and adulterated, following which the Provincial Quality Control Board directed to recall these medicines from the market.
These medicines include:
Injection Neudex 1mL. Dexamethasone sodium phosphate eq. to Dexamethasone phosphate 4mg/mL) Reg #042943
Batch No DX063, DX079: Expiry OCT 2027 and Batch No DX080: Expiry NOV 2027.
The above-mentioned injection was declared adulterated by Drug Testing Laboratories, Punjab.
The Directorate also recalled the following medicines, declaring them substandard based on Sterility Test.
Infusion Zeesol H 1000ml. (Ringer Lactate) Each 1000mL. contains: Calcium Chloride 2H2O: 0.27gms, Potassium Chloride: 0.40gms, Sodium Chloride: 6.00gms, Sodium Lactate: 3.20gms, Water for Injection: qs 2503281, Batch No 2503281, Expiry Feb-2030.
The Directorate also ordered the manufacturers of the aforementioned products to recall the affected batches from the market and furnish complete distribution trail, upload the “Recall Assessment Form” on the DRAP website, conduct a detailed root cause analysis, and prepare a Corrective and Preventive Action (CAPA) plan.
The provincial Drug Control Directorate also advised Healthcare Professionals & Regulators, Retailers (Pharmacies, Medical Stores), wholesalers, distributors, and healthcare facilities to stop dispensing of these products immediately and report the current inventory and consumption records to the respective area Drug Inspector/Drug Inspector Distributor.
All field formation in Punjab is also directed to enhance the surveillance within the supply chains and take regulatory action, in accordance with the applicable laws/rules as per Drug Act 1976/DRAP Act 2012.